The Canine Diversity Project
| Genetic Testing: A Guide for Breeders |
by Mary Whiteley, Ph.D.
By now today's breeders have heard about genetic testing, and DNA, but what is really involved in finding a mutation, and what do the results of the test mean?
What is a gene?
DNA (deoxyribonucleic acid) is a large molecule comprised of four basic units linked together in a long strand. If we assign each unit a letter, then we can envision the chromosome as a long string of letters. Genes can be thought of as long words scattered along the chromosome. These words have special meaning to the worker molecules in the cell, and encode a set of instructions as to what they are to build. Any mistake in a word scrambles the instructions, and either the workers in the cell make no product or a defective one is made. These mistakes in genes are known as mutations.There are basically two kinds of mutations, dominant and recessive. Every cell in the body (except sperm and eggs) has two copies of each gene (one from the sire, and one from the dam). A dominant mutation is where one defective copy of a gene is enough to affect the individual, whereas a recessive mutation requires that both copies of the gene carry a mutation for the individual to be affected.
How do scientists go about finding a mutation?
At the present time, there are two ways to identify mutations that cause genetic diseases. The first way is to find canine "homologs" of genes that cause a similar diseases in another species, such as mouse, rat or human. Genes involved in fundamental biological processes show a large degree of similarity between species. Scientists can take advantage of this conservation to "clone" (isolate) canine genes that may carry similar defects. Inbred strains of mice provide a wealth of information as to where to look in dogs. Likewise the human genome project, whose goal is the unlock the order of each letter along every chromosome, will also be invaluable in canine genetic research.Both the wild type and the suspected mutant genes need to be cloned and compared. In the case of PRA in Irish Setters, the culprit gene (rd1) was first identified in mice that had PRA. The defective "letter" in the mouse gene was not the same as in the Irish Setter, but it was the same gene, and the result was PRA in both species.
Direct tests
In the case of a discovery of an exact mutation, the diagnosis is accurate. For genes with one letter changes there is a simple way determine the presence of mutations by using diagnostic enzymes (called restrictions enzymes) that recognize a string of letters representing the region around the mutation, and the wild type sequence at the site of the mutationThe portion of the gene surrounding a mutation can be synthesized readily in the laboratory by a process called PCR (polymerase chain reaction). This method allow specific regions of the genome to be amplified from a small sample, and the DNA can be analyzed quickly. DNA samples for individuals can be obtained from any cells. They are most easily obtained by scrapping a few cells from the inner cheek with a small brush. All the ingredients required to make more DNA are put in a test tube and the DNA is made in a PCR machine. The key is the addition of a string of letters that corresponds to your gene to "prime" the synthesis.
Next, the amplified DNA is purified, and then cut with restriction enzymes. Two enzymes should be used for diagnosis, one for the wild type sequence, and one for the mutant sequence.
In this diagnosis, if you cut a wild type chromosome with an enzyme that recognizes the wild-type sequence, you get two pieces of DNA from the original one. They can be separated according to size on a gel matrix, and observed under ultraviolet light.
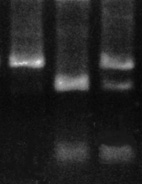
| If the animal is free of the mutation you will see only two pieces of DNA. If however, one of the chromosomes carries a mutation, you will see three fragments of DNA, the two from the wild-type chromosome, and a third, larger piece that was not cut by the enzyme. In the example shown here, the left lane contains uncut DNA, while the middle and right lanes have been cut with an enzyme that recognizes the wild-type sequence. The middle lane shows the two bands expected from a dog that is clear, while the right lane shows that dog to be a carrier (3 bands). An affected dog would give only a single band. |
Other defects may involve deletions of one letter or more in the gene. Diagnosis in this case would involve PCR amplification and the identification of the differently sized products. Digestion of these products with restriction enzymes which should give a predicted size pattern of DNA fragments. Additional fragments would be observed in carriers.
Linkage tests
The second, and less accurate way to identify mutations is known as linkage. Scattered throughout the chromosomes there exists short repeated groups of letters known as microsatellites (for example CACACACACACA). These can vary in length of repeat from individual to individual and are therefore referred to as simple sequence length polymorphisms (SSLP). Hundreds of these sequences have been isolated for the canine genome as tools for mapping genes. Because SSLPs can vary in length between individuals, they can be used to track defective genes. In order to find a microsatellite locus that is "linked" to a trait, you need a "family" of dogs in a pedigree. The disease status of dogs within this pedigree is made by some biochemical means or by physical examination depending on the defect. For example in the case of copper toxicosis in Bedlington terriers, the animals were determined as affected or unaffected by a liver biopsy, and a quantitative copper assay (Yuzbasiyan-Gurkan, et al., AJVM, 58:23-27, 1997). Knowing the status of the dogs then allows scientist to look for a microsatellite locus that is "linked" to the presence of disease. Hundreds of markers must be examined before a linkage with disease to a microsatellite is found.A linked microsatellite is said to co-segregate with the gene. The closer that the marker is linked to the disease, the more accurate the test. This needs to be reproduced with a goodly number of family members. Thus to find a gene with this method is relatively labor intensive.
Here is how the products of a PCR amplification of an SSLP closely linked to a gene for some genetic disease would look like when separated on a gel matrix.
Affected Non-affected Carrier ______ ______ ______ ______
In an affected animal, both copies of the microsatellite (one from each chromosome) are the same (they happen to be longer in this example) and both form the non-affected individuals are shorter. You must establish a correlation between the larger or smaller version of the microsatellite and the non-affected individual. In another family, the smaller version might be found in the affected animals because chromosomes can recombine, and the long microsatellite sequence may have crossed over to the chromosome carrying the wild type.
Because of this, the genotype of animals cannot be determined with absolute certainty. Accuracy is generally about 95-99%.
Performing this test is then trivial. It involves isolating DNA from an animal, synthesis of DNA using "primers" to a given microsatellite locus, and separating the products through a gel matrix. Because contamination is an ever-present risk, negative controls need to be included in the diagnostic experiment.
Because the isolation of a linked microsatellite identifies a specific region of the genome, it narrows down where to look for a specific gene causing a disease, and thus could ultimately lead to a specific test.
What the future holds
The area of genetic testing in dogs is so active that the prediction is that within a decade that there will be genetic tests for most of the genetic diseases in dogs. As well, one can envision tests for behavioral traits. One important note to breeders is that information must be used carefully, to make proper decisions for breeding in order to maintain genetic diversity in the breed. In the case of recessive disorders, if a carrier is bred to a clear, none of the puppies will be affected; however there is a 50% chance that individuals will be carriers. Likewise, there is a 50% chance that the puppies will be normal.Litters that are DNA-tested can be used to gradually eliminate the problem from the breed, without disturbing the gene pool.
© Mary Whiteley, 1997
Return to The Canine Diversity Project